Product Description and Product Data
This is an all-inclusive cell-based luciferase reporter assay kit targeting the the Human c-MET / Hepatocyte Growth Factor Receptor (c-MET/HGFR). INDIGO’s c-MET/HGFR reporter assay utilizes proprietary mammalian cells that have been engineered to provide constitutive expression of the OXTR. In addition to c-MET/HGFR Reporter Cells, this kit provides two optimized media for use during cell culture and in diluting the user’s test samples, a reference agonist, Luciferase Detection Reagent, and a cell culture-ready assay plate. The principal application of this assay is in the screening of test samples to quantify any functional activity, either agonist or antagonist, that they may exert against c-MET/HGFR. This kit provides researchers with clear, reproducible results, exceptional cell viability post-thaw, and consistent results lot to lot. Kits must be stored at -80C. Do not store in liquid nitrogen. Note: reporter cells cannot be refrozen or maintained in extended culture.
Features
Clear, Reproducible Results
- All-Inclusive Assay Systems
- Exceptional Cell Viability Post-Thaw
- Consistent Results Lot to Lot
Product Specifications
| Target Type | Growth Factor Receptor | ||
| Species | Human | ||
| Receptor Form | Hybrid | ||
| Assay Mode | Agonist, Antagonist | ||
| Kit Components |
| ||
| Shelf Life | 6 months | ||
| Shipping Requirements | Dry Ice | ||
| Storage temperature | -80C |
Data
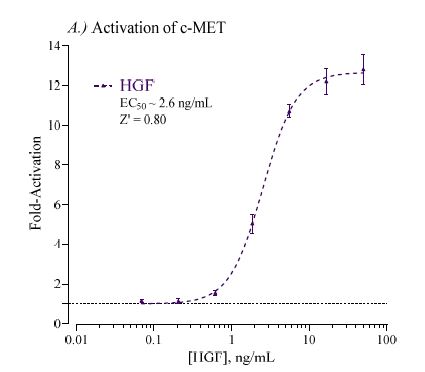

Target Background
The MET proto-oncogene encodes the receptor tyrosine kinase (RTK) c-MET, a.k.a Hepatocyte Growth Factor Receptor (HGFR). The c-MET receptor is formed by proteolytic processing of its precursor protein in the post-Golgi compartment into a singlepass, disulfide-linked α/β heterodimer. This cell surface receptor is expressed in cells of many organs, including the liver, pancreas, prostate, kidney, muscle, and bone marrow.
The only known ligand for c-MET is Hepatocyte Growth Factor (HGF). HGF acts as a pleiotropic factor and cytokine, promoting cell proliferation, survival, motility, differentiation and morphogenesis. The mature form of HGF consists of an α- and β- chain, which are held together by a disulfide bond. HGF binding to c-MET results in receptor homodimerization and phosphorylation of two tyrosine residues located in the intracellular tyrosine kinase domain.
At present, many studies have implicated c-MET in the regulation of cancer cell growth, angiogenesis, invasion and metastasis3. Deregulation and the consequent aberrant signaling of c-MET may occur by different mechanisms including gene amplification, overexpression, activating mutations, and increased ligand-mediated paracrine and autocrine stimulation. It has been established that c-MET is overexpressed in a variety of cancers including Lung, breast, ovary, kidney, colon, thyroid, liver, and gastric carcinomas. Consequently, c-MET and its ligand HGF continue to command much interest as targets for drug development and drug safety screening.
Citations
Also available as a service