ADME and Safety Pharmacology Assay Kits
Quickly Evaluate Therapeutic Candidates for ADME Properties and Safety Risks. INDIGO’s ADME/Safety Pharmacology assay kits enable you to rapidly assess potential drug-drug interactions, predict metabolic stability, and identify safety liabilities such as hepatotoxicity and other off-target effects.
These kits deliver fast, reliable data for:

Showing 1–10 of 21 results
-
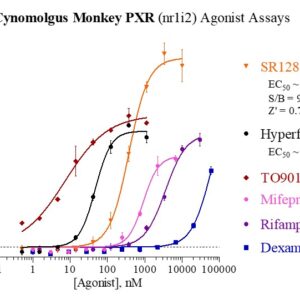
Cyn Monkey PXR Reporter Assay Kit
Species
Cyn Monkey
Target Type
Nuclear Hormone Receptor
Nuclear Receptor Orthologs
Associated Disease
Cancer, Cardiovascular, Kidney Disease, NASH/NAFLD, Wound Healing
-
CYP450 Induction Assay Kit
Species
Human
This CYP450 Induction Assay Kit contains optimized reagents for the culturing and treatment of semi-immortalized primary liver cells to assess drug-induced changes in CYP3A4, CYP1A1, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP2E1.
The kit provides two aliquots of semi-immortalized primary liver cells, two cell culture-ready assay plates, optimized Cell Culture Medium for use in all steps of the assay procedure, and three reference compounds (rifampicin, β-naphthoflavone, and CDCA) that activate one or more of the primary xenobiotic-sensing receptors: PXR, CAR, AhR, and FXR. Upon activation, these nuclear receptors modulate the expression of the CYP genes. Also included are seven sets of validated qPCR primers for quantifying drug-induced changes in the expression of CYP3A4, CYP1A1, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP2E1, as well as primers for ACTB.
Please note: This kit does not include reagents or protocols for cell lysis, RNA isolation, cDNA preparation, or qPCR assays.
-
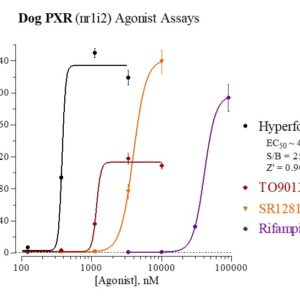
Dog PXR Reporter Assay Kit
Species
Dog
Target Type
Nuclear Hormone Receptor
Nuclear Receptor Orthologs
Associated Disease
Cancer, Cardiovascular, Kidney Disease, NASH/NAFLD, Wound Healing
-
Hepatotoxicity Assay Kit
Species
Human
The principal application of this assay is to rapidly screen test compounds to identify those that induce acute liver cell toxicity.
This kit includes two aliquots of cryopreserved Luminescent Reporter Hepatocytes (upcyte®), donor 10-13, isolated from an adult Caucasian female, that have been further modified to constitutively express the luciferase enzyme. In addition to two aliquots of the reporter hepatocytes, the kit provides two cell culture-ready assay plates, optimized Cell Culture Media (CCM) for use in all steps of the assay procedure (cell thawing, seeding, and preparation of the treatment media), luciferase detection reagent, and a reference compound that provides a positive control for hepatotoxicity.
The reagents and materials provided in this assay kit are formatted to allow the user to choose between two alternative assay setups. In one scenario 48 culture wells may be setup at two different times. In the other assay scenario 96 culture wells may be setup at one time.

-
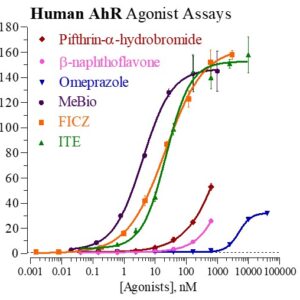
Human AhR Reporter Assay Kit
Species
Human
Target Type
Nuclear Hormone Receptor
Associated Disease
Cancer, Cardiovascular, Dermatitis, Kidney Disease, NASH/NAFLD, Reproduction, Wound Healing
-
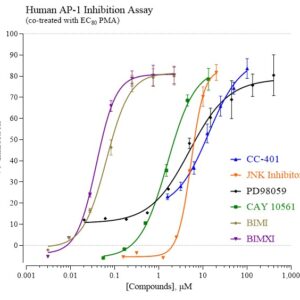
Human AP-1 Reporter Assay Kit
Species
Human
Target Type
Transcription Factor
Associated Disease
Cancer
-
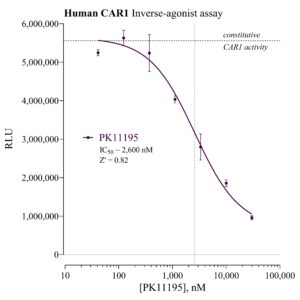
Human CAR-1 Reporter Assay Kit
Species
Human
Target Type
Nuclear Hormone Receptor
Associated Disease
NASH/NAFLD, Wound Healing
-
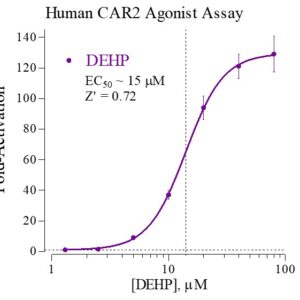
Human CAR-2 Reporter Assay Kit
Species
Human
Target Type
Nuclear Hormone Receptor
Associated Disease
NASH/NAFLD, Wound Healing
-
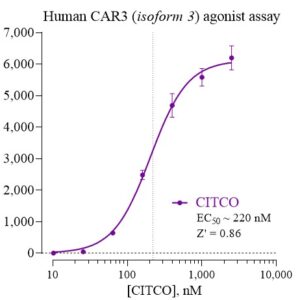
Human CAR-3 Reporter Assay Kit
Species
Human
Target Type
Nuclear Hormone Receptor
Associated Disease
NASH/NAFLD, Wound Healing
-
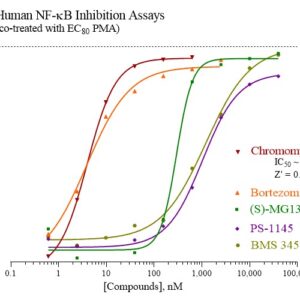
Human NF-κB Reporter Assay Kit
Species
Human
Target Type
Transcription Factor
Associated Disease
Autoimmune, Cancer, Cardiovascular, Dermatitis, Kidney Disease, Neurodegenerative