Product Description and Product Data
This Aryl Hydrocarbon Receptor bioassay kit for environmental sample monitoring is an all-inclusive luciferase reporter assay system that includes, in addition to AhR Reporter Cells, two optimized media for use during cell culture and in diluting the user’s test samples, Luciferase Detection Reagent, a cell culture-ready assay plate. and a detailed protocol.
Features
Clear, Reproducible Results
- All-Inclusive Assay Systems
- Exceptional Cell Viability Post-Thaw
- Consistent Results Lot to Lot
Product Specifications
| Target Type | Nuclear Hormone Receptor | ||
| Species | Mouse | ||
| Receptor Form | Native | ||
| Assay Mode | Agonist, Antagonist | ||
| Kit Components |
| ||
| Shelf Life | 6 months | ||
| Orthologs Available | Yes | ||
| Shipping Requirements | Dry Ice | ||
| Storage temperature | -80C |
Data

Target Background
The aryl hydrocarbon receptor (AhR) is a ligand-activated member of the basic helix loop helix- PER-ARNT-SIM (bHLH-PAS) family of transcription factors. It was initially characterized by its ability to sense xenobiotics, particularly environmental contaminants including 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD, also known as dioxin) and polycyclic hydrocarbons (PAHs), such as benzo[a]pyrene. Recent interest in AhR has also focused on its diverse physiological functions in a variety of systems, including but not limited to the liver, intestine, skin, and immune system.
The prototypical signaling pathway of AhR is similar to that of the nuclear receptor superfamily. In the absence of ligand, AhR is located in the cytoplasm, bound to a chaperone complex including a dimer of Hsp90, AIP, and p23. When bound to a ligand, the AhR translocates into the nucleus, where it binds to its heterodimerization partner, ARNT. The AhR-ARNT complex then binds specific cognate DNA sequence elements known as dioxin/xenobiotic/AHR response elements (DRC/XRE/AHRE), located upstream of responsive genes, most notable of which are members of the cytochrome P450 family of phase I drug metabolizing enzymes (predominantly CYP1A1, CYP1A2, and CYP1B1).
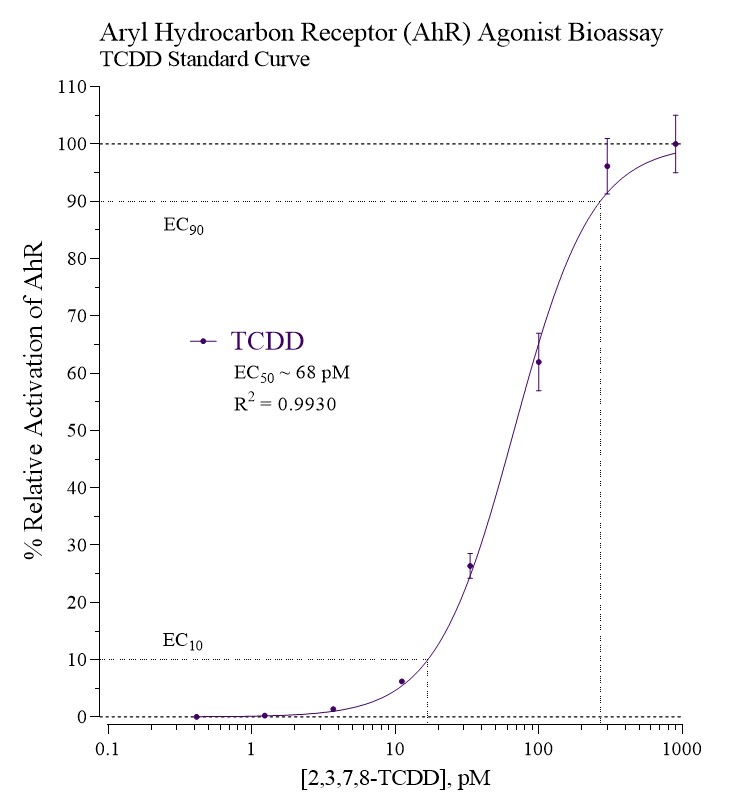
Differences in relative affinity for TCDD between species are known to exist. For example, the mouse AhRb-1 allele binds TCDD with 10-fold higher relative affinity than human AhR.3 For this reason, mouse AhR can be used as a sensitive tool for detecting the presence of dioxins and similar environmental pollutants.
INDIGO’s Mouse Aryl Hydrocarbon Receptor (AhR) Reporter Cells include the luciferase reporter gene functionally linked to an AhR-responsive promoter. Thus, quantifying changes in luciferase expression in the treated reporter cells provides a sensitive surrogate measure of the changes in mouse AhR activity.
The principal application of this assay is in the screening of test samples to quantify any functional activity, either agonist or antagonist, that they may exert against mouse AhR.
Product Documentation
Citations
Also available as a service